ROLE OF ATP
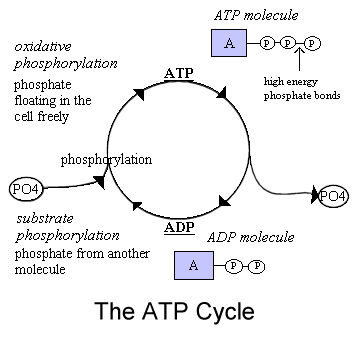
In order for the body to have energy, it must change the energy from the food into the form of energy our body likes to use, namely ATP, which stands for adenosine triphosphate. To produce ATP, our body cells use oxidation-reduction reactions, so let's take a look at how this works.
The hydrolysis of adenosine triphosphate (ATP) to adenosine diphosphate (ADP) and phosphoric acid (Pi) releases energy (it is an exergonic reaction). Some chemical reactions that occur in cells require energy.
This requires energy, and one way of providing this is from the oxidation of glucose which is an exergonic reaction.
There are two reasons why energy from the oxidation of glucose is not used directly to drive chemical reactions in the cell:
There are two reasons why energy from the oxidation of glucose is not used directly to drive chemical reactions in the cell:
- the hydrolysis of ATP releases small amounts of energy compared to the oxidation of glucose, and in a controlled way
- energy is released instantaneously from the hydrolysis of ATP, but the oxidation of glucose takes time