Carbohydrates are the most abundant biomolecule on Earth. Carbohydrate is a organic compound, it comprises of only oxygen, carbon and hydrogen. The ratio of hydrogen twice that of carbon and oxygen. The empirical formula being Cm(H2O)n (where m can be different from n). Carbohydrates include sugars, starches, cellulose and many other compounds found in living organisms. In their basic form, carbohydrates are simple sugars or monosaccharides. These simple sugars can combine with each other to form more complex carbohydrates. The combination of two simple sugars is a disaccharide. Carbohydrates consisting of two to ten simple sugars are called oligosaccharides, and those with a larger number are called polysaccharides.
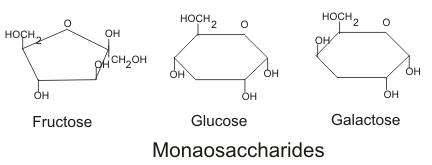
1. Monosaccarides
- Also called as simple sugars that are composed of 3-7 carbon atoms.
- Common base unit of all carbohydrate molecules.
- They have a free aldehyde or ketone group, which acts as reducing agents and are known as reducing sugars.
- Soluble in water.
- Sweet taste.
- Crystalline solid.
- Common monosaccarides: Glucose, galactose and fructose.
- Also called as simple sugars that are composed of 3-7 carbon atoms.
- Common base unit of all carbohydrate molecules.
- They have a free aldehyde or ketone group, which acts as reducing agents and are known as reducing sugars.
- Soluble in water.
- Sweet taste.
- Crystalline solid.
- Common monosaccarides: Glucose, galactose and fructose.
- Contain a single aldehyde or ketone functional group. They are subdivided into two classes aldoses and ketoses on the basis of whether they are aldehydes or ketones.
-They are also classified as a triose, tetrose, pentose, hexose, or heptose on the basis of whether they contain three, four, five, six, or seven carbon atoms.
-They are also classified as a triose, tetrose, pentose, hexose, or heptose on the basis of whether they contain three, four, five, six, or seven carbon atoms.
- Triose sugars: 3-carbon skeleton
- Pentose sugars: 5-carbon skeleton
- Hexose sugars: 6-carbon skeleton
2. Disaccarides
- Made of two monosaccharides.
- The bonds shared between two monosaccharides is the glycosidic bonds.
- A dehydration synthesis reaction forms a bond between carbon atoms in two monosaccharides, sandwiching an oxygen atom between them and releasing a water molecule.
- Soluble in water.
- Sweet taste.
- Crystalline solid.
- Common disaccarides: Sucrose, maltose and lactose.
- Made of two monosaccharides.
- The bonds shared between two monosaccharides is the glycosidic bonds.
- A dehydration synthesis reaction forms a bond between carbon atoms in two monosaccharides, sandwiching an oxygen atom between them and releasing a water molecule.
- Soluble in water.
- Sweet taste.
- Crystalline solid.
- Common disaccarides: Sucrose, maltose and lactose.
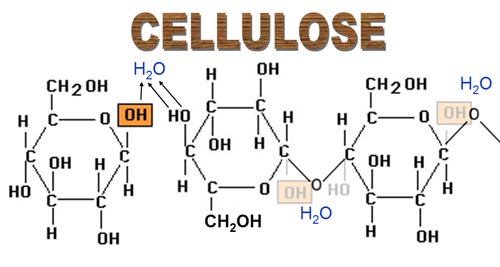
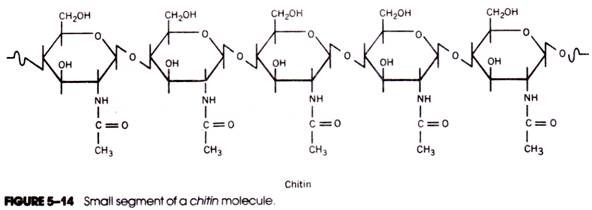
3. Polysaccarides
- Polymers of monosaccarides, carbohydrate chains with hundreds or more monosaccharide units.
- Complex carbohydrates.
- Unsweet.
- Insoluble in water.
- Not in crystalline form.
- Play vital energy storage and structural roles in living organisms, making carbohydrates the most abundant biomolecules on Earth.
- Common polysaccarides: Starch, cellulose, glycogen, pectin and dextrin.
- Polymers of monosaccarides, carbohydrate chains with hundreds or more monosaccharide units.
- Complex carbohydrates.
- Unsweet.
- Insoluble in water.
- Not in crystalline form.
- Play vital energy storage and structural roles in living organisms, making carbohydrates the most abundant biomolecules on Earth.
- Common polysaccarides: Starch, cellulose, glycogen, pectin and dextrin.